What “The Science” Really Says about the Coronavirus Pandemic
/The answer is not much – at least, not yet.
While advocates of lockdowns and masking mandates claim to be invoking “the science,” science by its very nature can’t provide short-term answers to the efficacy of such measures. The scientific method demands extensive data gathering and testing, which generally take longer than the duration of a pandemic. An abundance of scientific evidence does exist for the effectiveness of vaccination, but whether vaccines can completely eradicate the coronavirus is an open question. Social distancing as a preventive measure is also on firm scientific ground.
Lockdowns have been used for centuries as a way to slow the spread of disease, including the Black Death plague in the 14th century and the Spanish Flu in 1918-1919. But all they do is initially reduce transmission of the virus, and to claim otherwise is scientifically disingenuous.
The primary purpose of slowing down the spread of a contagious and deadly disease is to prevent the healthcare system from becoming overwhelmed. If more people get sick enough to require hospitalization than the number of hospital beds available, some won’t get adequate treatment and deaths will increase. However, lockdowns also have a devastating effect on a society’s economic and mental health. Studies have shown that negative socioeconomic impacts greatly limit the effectiveness of lockdowns over time.
In some countries such as Taiwan and Australia, death rates from COVID-19 are very low so far after repeated lockdowns, causing lockdown supporters to link the two. But other nations with small populations such as Israel have much higher mortality rates despite continued shutdowns. So there’s no correlation and, in fact, many other factors influence the death rate.
The science behind masking, shown below in use during the Spanish Flu pandemic, is muddier yet and has been badly contaminated by politics. Unfortunately, the gold standard in medical testing – the RCT (randomized controlled trial) – isn’t the basis for evaluating the benefit of mask-wearing by institutions like the U.S. CDC (Centers for Disease Control and Prevention) or the WHO (World Health Organization).
In an RCT or clinical trial, participants are divided randomly into two identical groups, with intervention in only one group and the other group used as a control. Neither the researchers nor the participants are told which group the participants are part of until the very end. Such double-blind trials are therefore able to establish causation.
For masks, just 14 RCTs have been carried out across the world to study how well masks guard against respiratory diseases, primarily influenza. Nearly all the trials tested so-called surgical, three-ply paper masks, rather than the N95 respirator style. Of the 14 trials, just two investigated the claim that wearing a mask benefits others who come in close contact with the mask wearer, while the other 12 tested the combination of benefit to others and protection for the wearer.
A recent analysis by a prominent statistician of all 14 RCTs, which include the only trial to test mask-wearing’s specific effectiveness against COVID-19, reveals that masks have no significant effect on either the wearer or those in close proximity, although some trials were ambiguous. There was no strong evidence that N95 masks performed any better than surgical or cloth masks. Exactly the same conclusions were reached in two independent analyses of 13 (see here) and 11 (here) of the same RCTs.
The CDC, however, relies on observational studies conducted since the start of the coronavirus pandemic, not RCTs, in issuing its masking guidance. An observational study is less scientific in being unable to assign a cause to an effect; it can only establish association.
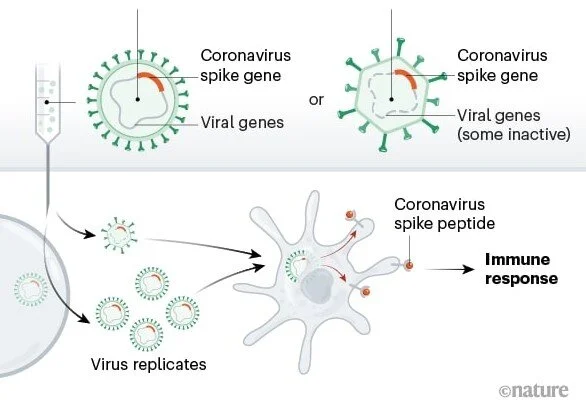
Vaccination against infectious diseases, on the other hand, has a solid scientific basis. Pioneered by Edward Jenner at the end of the 18th century, vaccination has eradicated killer diseases such as smallpox and polio in many countries, and drastically curtailed others such as measles, mumps and pertussis (whooping cough).
Nevertheless, the science underlying vaccination against COVID-19 is incomplete. In the past it’s taken several years to develop a new vaccine, but the COVID-19 vaccines currently available were brought to market at lightning speed. Although such haste was seen as necessary to combat a rapidly proliferating virus, it meant shortening the RCTs designed to test vaccine efficacy, leaving questions such as long-term side effects and duration of effectiveness unresolved.
And barely understood yet is the greater protection against infection acquired though natural immunity – the result of having recovered from a previous COVID-19 infection – than from vaccination. This complicates calls for vaccine mandates, as those with natural immunity arguably don’t need to be vaccinated.
Moreover, the coronavirus is an RNA virus like influenza and so frequently mutates. This means that mandatory vaccination for the coronavirus is unlikely to be any more effective community-wide than a mandated flu vaccine would be. Regular COVID-19 booster shots will probably be needed, just like the flu.
That’s where science stands on the coronavirus. But rather than following the science, most decisions on lockdowns, masking and vaccination are ruled by politics.
Next: Sea Ice Update: No Evidence for Recent Ice Loss